Nowadays, tuberculosis is -aside with HIV and malaria- one of the three single leading causes of death at a global level; with a dramatic burden in underdeveloped countries of Africa and Asia. According recent WHO estimations, more than in 2015, up to 9.5 new million individuals developed the disease all around the world; and as many as 1.5 million people died for it. Its causative agent, –the bacillus Mycobacterium tuberculosis– is a human obligate parasite whose elusive life cycle involves many different spatial and temporal scales.
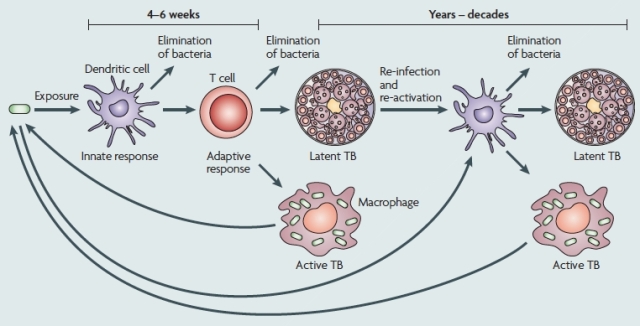
Tuberculosis infection process. After exposure to tuberculosis bacilli, depending on the strength of the immune response different outcomes are possible: from successful elimination of bacteria to prompt development of disease (primoinfection). A third outcome, around nine times more common than a fast progression to disease consists on immunological containment of dormant bacteria within granulomes, compatible with an asymptomatic latent state in the host that can last for her whole life. The figure is extracted from the work by Young, Stark and Kirschner: “Systems biology of persistent infection: tuberculosis as a case study“, published in Nature Reviews Microbiology in 2008.
Particularly, the enormous temporal gap often appearing between tuberculosis infection and active disease makes its mathematical description at populations level a subtle matter. Similarly, the absence of adequate immunological correlates of infection and protection makes the evaluation of any epidemiological intervention specially difficult, which constitutes a dramatic caveat that finds its truly origin at the immunological level of description, and our incomplete knowledge of it. In addition, another aspects make the pathogen highly interesting from a complex systems perspective. For example, the reduced levels of genetic diversity of the pathogen allow us to faithfully track its evolutionary history and to relate it to that of its host. Additionally, the highly difficult, slow and expensive wet lab protocols in tuberculosis research claim for the development of reliable in-silico approaches able to guide and facilitate experimental work. At this level, the generalization of high-troughput “omics” technologies have allowed the appearance of a large amount of experimental data at the level of single cells during the last years, which has derived in a deeper knowledge of the cell biology of the pathogen and its interactions with the host, and has opened the way for the development of more accurate models and quantitative tools.
All these features unveil the multiplicity of the conceptual, temporal and spatial scales involved in the process of tuberculosis infection; which ultimately make it an optimal target of a complexity research program.
![]() The contents in this website are licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
The contents in this website are licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.